A political retreat, an unanswered appeal, and states building their own guardrails
Welcome to The State of U.S. Vaccine Policy, our biweekly series in partnership with CIDRAP at the University of Minnesota. Two weeks ago, we came to you with a Special Edition celebrating the March 16th court ruling as a genuine win for vaccines, science, and the rule of law—and it still is. But as we said then, it was a preliminary ruling, not a final one.
Every fall, I scramble to get my garden hoses into storage before the first deep freeze. Like clockwork, every spring, I’m left with a tangled mess that needs to be sorted before I can actually use them again. The preliminary ruling was spring poking through—a reason for real optimism. But we’re still here holding a pile of tangled hoses.
Let’s discuss…
The PDF version of today’s update is here. Read past installments here and today’s update on the Unbiased Science Substack here.
Has Vaccine Policy Lost Its Midterm Appeal?
Something notable has shifted in Washington, DC. Heading into the midterms (officially now six months away), Health and Human Services (HHS) Secretary Robert F. Kennedy Jr.’s vaccine skepticism is becoming a political liability, and the administration appears to be quietly stepping back from it.
Robert Malone, who served as the Advisory Committee on Immunization Practices (ACIP) vice chair until recently (more on that shortly), said as much publicly. In a podcast episode with the Informed Consent Action Network (ICAN), he warned that the White House had made a strategic decision that the Make America Healthy Again (MAHA) coalition was an “inconsequential minority” that wouldn’t affect the midterms, and that the silencing of vaccine criticism was alienating the base. As he said, “How can you have Bobby [Kennedy] on Joe Rogan and the word ‘vaccine’ never escapes his lips? That’s a tell.”
Beyond Malone’s commentary, the pattern speaks for itself. The administration has not yet appealed the American Academy of Pediatrics (AAP) v. Kennedy ruling, and is still deciding how to proceed. Casey Means’ surgeon general confirmation has stalled. President Donald Trump failed to nominate a permanent director of the Centers for Disease Control and Prevention (CDC) by the statutory deadline. And the White House is actively redirecting its health policy toward food and chronic disease—topics for which MAHA polls far better than for vaccines. Four senior administration officials confirmed to Reuters this week that the pivot is deliberate.
So what does this all mean? A political retreat from vaccine skepticism is not the same as a true course correction. You can’t simply flip a switch and expect things to return to where they were, and we are far from rebuilding after the damage done over the past year. Our hoses remain woefully tangled.
Dizzying Personnel Chaos from the Revolving Door That Won’t Stop Spinning
We’ve been tracking the leadership vacuum across federal health agencies since this series began. If you’re having trouble keeping track (and who could blame you?), we’ve made you an org chart. Here’s where things stand:
Robert Malone’s ACIP Exit
As part of Judge Brian Murphy’s stay that was issued in the AAP v Kennedy case on March 16th, he found that the ACIP members had been improperly appointed. This ruling came just days before a scheduled ACIP meeting, leaving the agency scrambling. In the days that followed, Malone told the Wall Street Journal that he and other committee members had been informed that the committee would disband and then be remade. That night, he reversed course on X, citing it as a miscommunication.
That miscommunication appears to have been the beginning of the end for Malone. He resigned from his position as ACIP vice chair on March 24th and did not sugar-coat the frustration behind his decision. He told the New York Times that a feud with HHS staff following the ruling was his last straw, adding: “Hundreds of hours of uncompensated labor, incredible hate from many quarters, hostile press, internal bickering, weaponized leaking, sabotage… I have better things to do.”
Malone isn’t actually a fringe figure by background. He contributed foundational work on mRNA lipid delivery systems in the late 1980s, research that underpins the COVID-19 vaccines he spent much of the pandemic publicly attacking. His turn toward vaccine skepticism made him a prominent voice in MAHA circles, but his exit is yet another signal of just how chaotic things have become inside HHS, and a reminder that the retreat from vaccine skepticism is creating fractures even within MAHA, not just between MAHA and the broader medical community.
No New CDC Director
Following the departure of acting director Jim O’Neill earlier this year, HHS had a 210-day statutory deadline to nominate a new CDC director. That deadline was March 25th, and it came and went without a nomination. NIH Director Jay Bhattacharya will continue to serve as acting CDC director. He addressed CDC staff this week, acknowledging the difficulty of the past year and promising a return to stability, though it remains to be seen what this will look like in practice.
Finding the right candidate has proven rather difficult: the administration needs someone who aligns with the MAHA agenda, all while distancing itself from Kennedy’s less popular vaccine positions and clearing a Senate confirmation. It’s hard to overstate just how high the stakes are in getting this right. A recent STAT opinion piece by three senior physician-scientists argued that the CDC’s most urgent problem isn’t scientific capacity, but that public trust has completely collapsed. Without a director who is wholly committed to rebuilding that credibility, even scientifically sound recommendations likely won’t translate into action.
The short list of candidates reportedly includes Ernie Fletcher, a family physician and former Kentucky governor; Joseph Marine, a Johns Hopkins cardiologist with closer ties to the MAHA movement; and Daniel Edney, the Mississippi state health director. Individuals familiar with the internal process say there are roughly half a dozen people under consideration, and that President Trump could decide to go in a completely different direction altogether. (The suspense is killing us.)
FDA Vacancies Aren’t Faring Much Better
Applications for the new Center for Biologics Evaluation and Research (CBER) director—the Food and Drug Administration (FDA) division that oversees vaccine approvals—are due tomorrow, following Vinay Prasad’s resignation in early March. (That means you have just one day to apply here.) Separately, Adam Sherwat, director of the Office of Infectious Diseases at CDER, has announced his resignation with a last day of April 4th. It’s not readily clear why he’s leaving, but the bottom line is that two of the FDA’s most senior vaccine-adjacent roles are now simultaneously vacant.
On Casey Means
The status of Casey Means’ surgeon general nomination took some remarkable whiplash last weekend. When reporters asked President Trump this week directly whether he might withdraw her nomination, he said that “something like that would be possible,” adding that there were “a lot of great candidates.” The following day, the White House issued a formal statement praising Means’ background and calling on the Senate to confirm her without delay. The president and his press secretary sent completely contradictory messages within 24 hours.
The underlying problems with Means that became readily apparent at her February hearing haven’t gone away: Her position on vaccines, credentials, and broader posture toward established medical institutions left many unsatisfied, including several Republican senators. The inactive license issue has come to the forefront, too. Former Surgeon General Jerome Adams—who served under President Trump during the COVID-19 pandemic—has publicly argued that her inactive medical license isn’t just a stylistic concern, but a functional disqualifier. The fact that it’s President Trump’s own first-term surgeon general making this argument and telling reporters that Republicans in Congress privately share his concerns makes it harder to dismiss.
When NBC News asked HELP Committee Chairman Senator Bill Cassidy directly about a timeline for Means’ vote on Tuesday, he said “no comment.” Hours later, the Senate adjourned for a two-week Easter recess, which means there will not be a committee vote until mid-April at the earliest.
AAP v. Kennedy: What We’re Seeing (And What We’re Not)
Children’s Health Defense (CHD) has announced it will appeal both of Judge Murphy’s rulings: his February 27th denial of CHD’s motion to intervene, and the March 16th stay that paused vaccine policy changes and blocked further ACIP activity. CHD filed an emergency motion in the First Circuit on March 24th, seeking expedited consideration and injunctive relief pending the appeal.
Things aren’t quite as straightforward at the federal level, and, honestly, the delay in action is notable. Historically, the Trump administration has moved quickly to appeal other court rulings that it disagreed with, making two weeks of inaction here a bit conspicuous. HHS spokesperson Andrew Nixon has continued to say only that any assertions about next steps are “baseless speculation” until officially announced.
Let’s talk about the options that are on the table. According to Reuters, one option is a narrow appeal focusing specifically on the ACIP member appointments rather than the full ruling. A second is accepting the ruling entirely and reconstituting the ACIP through the proper legal processes, though that path comes with its own catch: The vetting process for new members typically takes at least a year, which means that fall vaccine recommendations could be in jeopardy even under the most cooperative scenario. The third option—and the one that MAHA allies seem to be pushing for—is a broad appeal to the First Circuit, which is required before the case could proceed to the Supreme Court. The Justice Department has a 60-day window to file. For a broader perspective on what the ruling does and doesn’t resolve, CIDRAP’s Vaccine Integrity Project viewpoint piece is worth reading; a legal win is not a reason to stand down. And regardless of which option the federal government chooses, rest assured it will be a calculated move leading up to the midterm elections.
If the ACIP were to be reconstituted, it would likely be asked to vote on new ICD-10 billing codes for post-COVID vaccine injuries and long COVID, neither of which is within ACIP’s authority. Those codes are maintained by the Centers for Medicare and Medicaid Services (CMS) and the National Center for Health Statistics, so any vote would be non-binding. It’s also important to keep in mind that ICD-10 codes reflect what a clinician documents at billing, but are not confirmed causation.
State Vaccine Policy: Saved By the Bell
The past two weeks have seen a meaningful cluster of pro-vaccine actions at the state level, and it’s worth noting that they cross party lines. Meanwhile, many state legislatures are winding down for the year. In the map at left, states whose legislative sessions end in March are colored red, and legislative sessions ending in April are green.
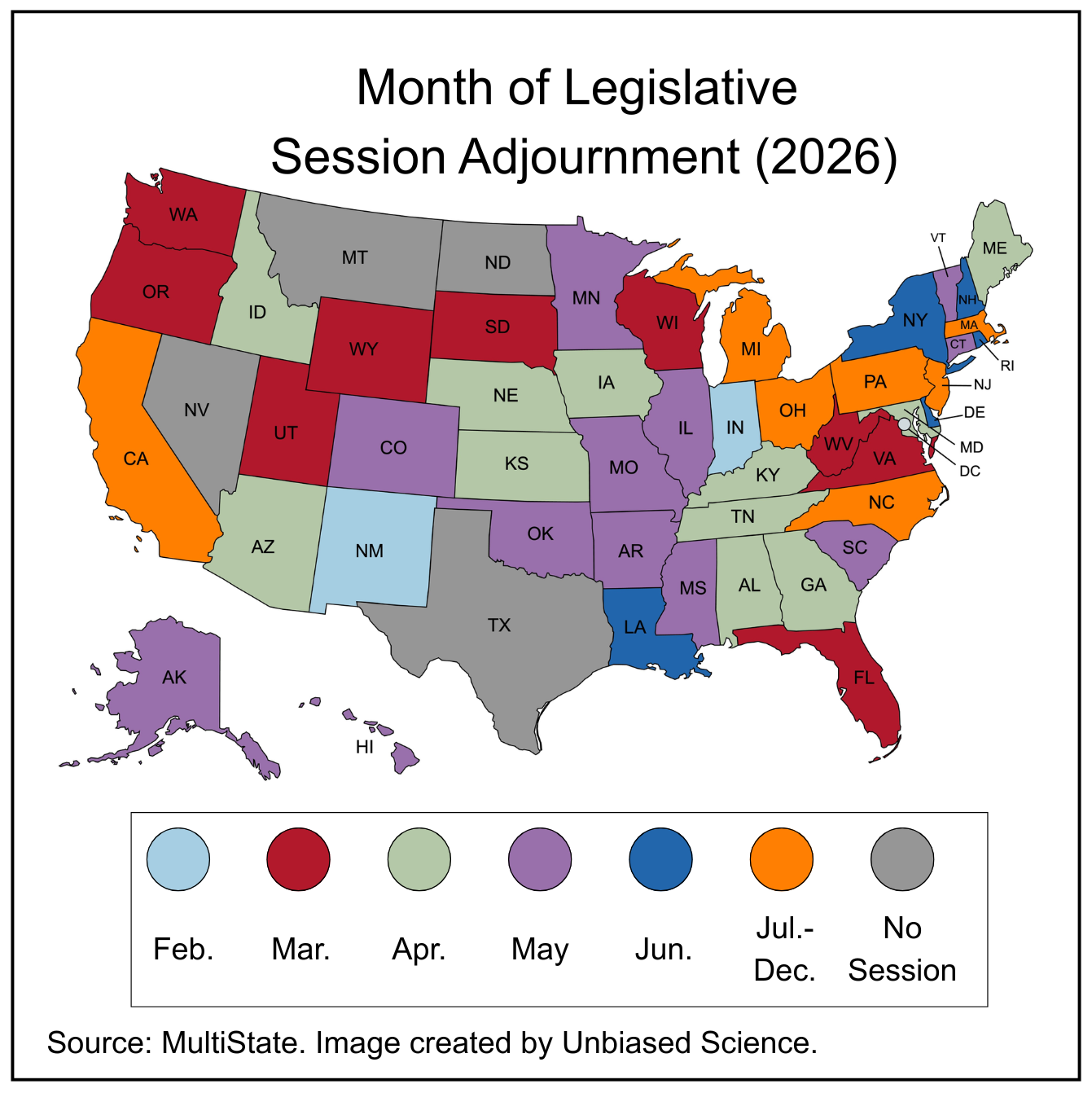
Iowa’s bill to remove all school vaccine requirements failed to meet the second legislative funnel deadline in mid-March and will not advance this session, which is slated to adjourn on April 21st.
Colorado Governor Jared Polis signed SB 26-32 into law last Friday, completing what we noted in our last issue as one of the most comprehensive pro-vaccine bills in the country. The law allows the State Board of Health to consider vaccine guidance from the AAP and other major medical organizations (not just the CDC), codifies pharmacists’ authority to prescribe and administer vaccines, and allows the state to use general fund money to purchase childhood vaccines if the funding for the federal Vaccines For Children program is cut.
In Vermont, Republican Governor Phil Scott signed H.545 on March 24th, enabling the Vermont Department of Health to make vaccine recommendations based on available science and data, independent of federal guidance.
A two-bill package was introduced in New York on March 27th, with support from the governor, senator majority leader, and assembly speaker. The first bill would require health insurers to cover the cost of any vaccines recommended by the state health commissioner, in addition to those recommended by the ACIP. The second would remove explicit references to the ACIP from state public health, education, and Medicaid law, and expand pharmacists’ authority to administer COVID-19 vaccines to children aged 2 to 18 years. These are bills, not yet law, but the fact that all three branches of state government are aligned behind them signals serious intent.
We first covered Oregon’s SB 1598 in our March 5th issue, and it was signed into law this week. Briefly, it requires insurance plans to continue covering vaccines with no out-of-pocket costs and allows the state’s public health officer to recommend covered vaccines independently of federal guidance.
The Common Health Coalition released the Model State Legislation: Vaccine Provider Protection Act—a template that states can use to protect physicians from liability and political pressure when recommending vaccines in line with professional guidelines.
Keep an eye on New Hampshire today. The House Health and Human Services Committee is holding a hearing this morning on three bills that, if passed, would roll back existing vaccine protections in the state: HB 1719 would remove the hepatitis B vaccine from the list of vaccines required under state law, HB 1584 would require the department of health to provide notice of medical and religious vaccine exemptions, and; HB 1449 would both limit the times that vaccine clinics can operate at schools and require parents or guardians to be physically present during any vaccination. We’ll have updates in our next issue.
Here’s What Else We’re Watching:
- Vaccine Injury Table: Aaron Siri, a vaccine injury lawyer and long-time Kennedy ally, filed a petition on March 20th demanding that HHS add more than 300 new conditions to the Vaccine Injury Table, threatening a federal lawsuit if Kennedy doesn’t act within 60 days. The petition argues that the government’s mere consideration of a potential vaccine injury is sufficient grounds for adding it to the table, while legal experts have weighed in to say this is a significant stretch. This slid under the rug while the ACIP ruling occupied the headlines.
- Cancer Vaccines: The Trump administration may be heavily investing in cancer vaccines through a potential $200 million public-private partnership at the National Cancer Institute, focused on therapeutic vaccines for patients at high risk of cancer recurrence. Vaccines that treat disease rather than prevent it appear to occupy a different political space right now.
- Lyme Vaccine: At an HHS hearing on March 17th, Kennedy said, “There is nobody who will fight harder to find a vaccine or treatment for Lyme disease than me.” This is particularly important in light of Pfizer and Valneva’s announcement that their candidate Lyme vaccine showed 70% efficacy in a Phase 3 trial, though that trial came with some caveats.
- COVID-19 Vaccine Trial Halted: Pfizer and BioNTech just halted a large U.S. trial of their updated COVID-19 vaccine in healthy adults aged 50 to 64 because they were unable to recruit 25,000 to 30,000 people in that age range without chronic conditions such as hypertension and diabetes. Results from the study were going to be presented at the FDA’s Vaccines and Related Products Advisory Committee meeting in May to guide decisions on COVID-19 vaccine strain selection in the fall. Without the data, it’s possible that “there may not be a specific approval for this age group,” said former FDA chief scientist Dr. Jesse Goodman.

The work of keeping vaccines accessible and grounded in science isn’t always flashy—it can feel more like untangling garden hoses than tending a garden. But the untangling is what makes the garden possible. Thank you for doing it with us.
Stay curious,














